Embarking on the Basics: Exploring Units and Dimensions
Fundamental Physical Quantities in the SI System: World of Dimensional Formula Class11
-
- Defined by the International System of Units (SI).
- Include seven fundamental quantities: length, mass, time, electric current, temperature, amount of substance, and luminous intensity.
- Serve as the foundation for measuring other physical quantities.
- Expressed in terms of standard SI units such as meters, kilograms, seconds, amperes, kelvin, moles, and candelas.
Supplementary Physical Quantities in the SI System: World of Dimensional Formula Class11
-
- Additional quantities derived from fundamental ones to enhance measurement precision or describe complex phenomena.
- Examples include plane angle (radian), solid angle (steradian), and frequency (hertz).
- Plane angle and solid angle supplement length and provide a means to measure rotation and spatial extent in three dimensions.
- Frequency complements time, indicating the number of cycles or occurrences per unit time.
- While not fundamental, these supplementary quantities play vital roles in various scientific and engineering applications within the SI framework
Supplementary Quantities: World of Dimensional Formula Class11
-
- Augment the fundamental quantities in the International System of Units (SI) to provide a more comprehensive framework for measurement.
- Include plane angle, solid angle, and frequency.
- Plane angle, measured in radians, supplements length by quantifying rotation or angular displacement.
- Solid angle, measured in steradians, complements length by quantifying spatial extent in three dimensions, crucial in fields like optics and astronomy.
- Frequency, measured in hertz (Hz), supplements time by indicating the number of cycles or occurrences per unit time interval, pivotal in fields such as telecommunications, physics, and engineering.
- While not considered fundamental, supplementary quantities play essential roles in various scientific and engineering disciplines, enriching the SI system and enabling precise measurements and analyses of physical phenomena.

What Are Dimensions? World of Dimensional Formula Class11
What Are Dimensional Constants? World of Dimensional Formula Class11
- Speed of Light (c):
- The dimensional formula of speed is [LT^-1], representing length per unit time. The speed of light, being a velocity, has the same dimensional formula as speed.
- Therefore, the dimensions of the speed of light, denoted as [c], are [LT^-1].
- Planck’s Constant (h):
- Planck’s constant relates energy to frequency in quantum mechanics, as given by the equation 𝐸=ℎ𝑓, where E is energy, h is Planck’s constant, and f is frequency.
- Rearranging the equation to solve for h, we have ℎ=𝐸𝑓.
- Energy has the dimensional formula [ML^2T^-2], and frequency has the dimensional formula [T^-1].
- Substituting the dimensional formulae into the equation, we find that the dimensions of Planck’s constant, denoted as [h], are [ML^2T^-1].
- Gravitational Constant (G):
- The gravitational constant relates the force of gravity to the masses of objects and the distance between them, as described by Newton’s law of universal gravitation.
- Using Newton’s law, 𝐹=𝐺⋅𝑚1⋅𝑚2𝑟2, where F is force, G is the gravitational constant, 𝑚1 and 𝑚2 are masses, and r is the distance between the centers of masses.
- Rearranging the equation to solve for G, we have 𝐺=𝐹⋅𝑟2𝑚1⋅𝑚2.
- Force has the dimensional formula [MLT^-2], and distance has the dimensional formula [L].
- Substituting the dimensional formulae into the equation, we find that the dimensions of the gravitational constant, denoted as [G], are [L^3M^-1T^-2].
- Boltzmann Constant (𝑘𝐵):
- The Boltzmann constant relates the average kinetic energy of particles in a gas to the temperature of the gas, as given by the equation 𝐸=𝑘𝐵⋅𝑇, where 𝐸 is energy, 𝑘𝐵 is the Boltzmann constant, and 𝑇 is temperature.
- Rearranging the equation to solve for 𝑘𝐵, we have 𝑘𝐵=𝐸𝑇.
- Energy has the dimensional formula [ML^2T^-2], and temperature has the dimensional formula [Θ].
- Substituting the dimensional formulae into the equation, we find that the dimensions of the Boltzmann constant, denoted as 𝑘𝐵, are [ML^2Θ^-1].
- Avogadro’s Number (𝑁𝐴):
- Avogadro’s number represents the number of atoms, molecules, or particles in one mole of a substance.
- Since Avogadro’s number is a count of particles, it is dimensionless.
- Faraday Constant (𝐹):
- The Faraday constant represents the amount of electric charge carried by one mole of electrons, and it is used in electrochemistry.
- Charge has the dimensional formula [IT].
- Since the Faraday constant is a count of charge per mole, its dimensions are [IT N^-1].
- Rydberg Constant (𝑅):
- The Rydberg constant is used in atomic physics to describe the wavelengths of spectral lines of hydrogen.
- It is derived from the fundamental constants of nature and has dimensions of [T^-1], representing frequency.
- Gas Constant (𝑅):
- The gas constant relates the energy of a gas to its temperature and pressure, as given by the ideal gas law equation 𝑃𝑉=𝑛𝑅𝑇, where 𝑃 is pressure, 𝑉 is volume, 𝑛 is the number of moles, 𝑇 is temperature, and 𝑅 is the gas constant.
- Rearranging the equation to solve for 𝑅, we have 𝑅=𝑃𝑉𝑛𝑇.
- Pressure has the dimensional formula [ML^-1T^-2], volume has the dimensional formula [L^3], amount of substance has the dimensional formula [N], and temperature has the dimensional formula [Θ].
- Substituting the dimensional formulae into the equation, we find that the dimensions of the gas constant, denoted as 𝑅, are [ML^2T^-2Θ^-1N^-1].
- Stefan-Boltzmann Constant (𝜎):
- The Stefan-Boltzmann constant relates the total power radiated by a black body to its temperature, as described by Stefan-Boltzmann law equation 𝑃=𝜎𝐴𝜀𝑇4, where 𝑃 is power, 𝐴 is the surface area, 𝜀 is the emissivity, 𝑇 is temperature, and 𝜎 is the Stefan-Boltzmann constant.
- Rearranging the equation to solve for 𝜎, we have 𝜎=𝑃𝐴𝜀𝑇4.
- Power has the dimensional formula [ML^2T^-3], surface area has the dimensional formula [L^2], emissivity is dimensionless, and temperature has the dimensional formula [Θ].
- Substituting the dimensional formulae into the equation, we find that the dimensions of the Stefan-Boltzmann constant, denoted as 𝜎, are [ML^2T^-3Θ^-4].
- Molar Gas Constant (𝑅):
- The molar gas constant, also known as the universal gas constant, is defined as the product of the Boltzmann constant and Avogadro’s number, and it is used in thermodynamics.
- Since the Boltzmann constant has dimensions [ML^2Θ^-1] and Avogadro’s number is dimensionless, the dimensions of the molar gas constant, denoted as 𝑅, are [ML^2Θ^-1N^-1].
- Permittivity of Free Space ():
- The permittivity of free space, also known as vacuum permittivity, is a fundamental constant in electromagnetism. It represents the ability of a vacuum to permit the transmission of electric fields.
- Permittivity has the dimensions of [M^-1L^-3T^4I^2], representing electric charge squared divided by force multiplied by length squared.
- Therefore, the dimensions of the permittivity of free space, denoted as 𝜀0, are [M^-1L^-3T^4I^2].
- Permeability of Free Space ():
- The permeability of free space, also known as vacuum permeability, is a fundamental constant in electromagnetism. It represents the ability of a vacuum to permit the transmission of magnetic fields.
- Permeability has the dimensions of [MLT^-2I^-2], representing force per unit current squared.
- Therefore, the dimensions of the permeability of free space, denoted as 𝜇0, are [MLT^-2I^-2].
- Electric Conductivity (𝜎):
- Electric conductivity represents the ability of a material to conduct electric current. It is the reciprocal of resistivity.
- Conductivity has the dimensions of [ML^-3T^3I^2], representing current per unit electric field multiplied by length.
- Therefore, the dimensions of electric conductivity, denoted as 𝜎, are [ML^-3T^3I^2].
- Magnetic Permeability (𝜇):
- Magnetic permeability represents the ability of a material to respond to an applied magnetic field.
It is the reciprocal of magnetic susceptibility.
- Permeability has the dimensions of [MLT^-2I^-2], representing force per unit current squared.
- Therefore, the dimensions of magnetic permeability, denoted as 𝜇, are [MLT^-2I^-2].
Watch this Video-
Some Examples of Dimensionless Quantities: World of Dimensional Formula Class11
Here are some examples of dimensionless quantities:
- Reynolds Number: Used in fluid mechanics, it represents the ratio of inertial forces to viscous forces in a fluid flow, determining whether the flow is laminar or turbulent.
- Mach Number: In aerodynamics, it denotes the ratio of the speed of an object to the speed of sound in the surrounding medium, indicating whether the object is traveling at subsonic, transonic, supersonic, or hypersonic speeds.
- Euler’s Number (e): In mathematics, it is a fundamental constant representing the base of the natural logarithm. It arises in various mathematical contexts, such as compound interest calculations and exponential growth.
- Aspect Ratio: In geometry, it refers to the ratio of the length of the longer side of a shape to the length of its shorter side. It characterizes the shape’s elongation or compactness and is widely used in engineering, architecture, and design.
- Refractive Index: In optics, it signifies the ratio of the speed of light in a vacuum to the speed of light in a given medium. It determines how much light is bent or refracted as it passes from one medium to another, such as air to glass.
- Prandtl Number: Another dimensionless quantity in fluid mechanics, it represents the ratio of momentum diffusivity to thermal diffusivity in a fluid flow. It describes the relative importance of momentum and heat transfer in a fluid.
- Coefficient of Friction: In mechanics, it denotes the ratio of the force of friction between two surfaces to the force pressing them together. It quantifies the resistance to motion between the surfaces and is crucial in designing mechanical systems.
Law of Homogeneity of Dimensions: World of Dimensional Formula Class11
Checking the Dimensional Consistency
Checking the Dimensional Consistency involves verifying whether the dimensions of all terms in an equation or expression are compatible with each other. This process ensures that the equation is mathematically valid and physically meaningful.
For instance,
consider the equation for the period 𝑇 of a simple pendulum:
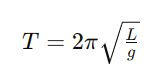
Where:
- 𝑇 is the period of the pendulum,
- 𝐿 is the length of the pendulum, and
- 𝑔 is the acceleration due to gravity.
To check the dimensional consistency of this equation, we assign dimensions to each term:
- The dimension of period 𝑇 is represented by [T] (time).
- The dimension of length 𝐿 is represented by [L] (length).
- The dimension of acceleration due to gravity 𝑔 is represented by [LT^-2] (length per time squared).
Substituting these dimensions into the equation:
Dimension of L.H.S = [
Dimension of R.H.S
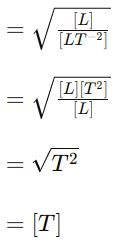
This confirms that the equation accurately represents the relationship between the period, length, and acceleration due to gravity of a simple pendulum.
Deducing the Relation among Physical Quantities using Dimensions: World of Dimensional Formula Class11
“Deducing the Relation among Physical Quantities using Dimensions” involves using the principle of dimensional analysis to derive relationships between different physical quantities based on their dimensions. This method is particularly useful when direct experimental measurements are unavailable or impractical.
Question:
![]()



FAQs: World of Dimensional Formula Class11
- Q: What is a dimensional formula?
- Answer: A dimensional formula represents the expression of a physical quantity in terms of its fundamental dimensions, such as length, mass, and time. It helps in understanding the nature of the physical quantity and its dependence on fundamental units.
- Q: How do you calculate dimensional formulae?
- Answer: To calculate the dimensional formula of a physical quantity, express it in terms of fundamental dimensions using its defining equation. Assign dimensions to each term in the equation and simplify to obtain the final dimensional formula.
- Q: Why are dimensional formulae important?
- Answer: Dimensional formulae play a crucial role in physics as they provide insights into the nature of physical quantities, help derive relationships among them, and facilitate dimensional analysis, which is essential for problem-solving and theoretical analysis.
- Q: Can dimensional analysis be used to check equations for correctness?
- Answer: Yes, dimensional analysis is a powerful tool for checking the correctness of equations. If the dimensions on both sides of an equation match, it indicates that the equation is dimensionally consistent and likely to be correct.
- Q: How are dimensional formulae applied in real-life scenarios?
- Answer: Dimensional formulae find applications in various fields such as engineering, chemistry, and physics. They are used to derive equations, predict physical behavior, design experiments, and analyze data, making them indispensable in scientific research and practical applications.
Also Read-
https://pcmconceptclear.com/best-book-for-science-class-11-cbse/
https://pcmconceptclear.com/best-science-book-for-class-11-isc/
https://pcmconceptclear.com/physics-class-11-units-and-measurement/
https://pcmconceptclear.com/some-basic-concepts-of-chemistry-cbse-11/
https://pcmconceptclear.com/mastering-mole-concepts-class-11/




9 thoughts on “World of Dimensional Formula Class11: Expert Tips(2025)”